The Science of a Kitchen Fire: Why Water Makes It Worse
A kitchen fire is a pool of cooking oil sitting a few degrees below its autoignition point. Here's the physics of why water makes it catastrophically worse.

Your kitchen ranks, statistically, as the most dangerous room in your house. In fact, a kitchen fire leads every other type of domestic fire in every industrialised country with comparable data. Moreover, the pattern has held steady for decades. Cooking causes roughly 44% of home structure fires in the United States, 44% of accidental dwelling fires in England, and the single largest share of domestic fires across Europe.
So why does a kitchen fire prove so hard to fight, and why does our instinct to grab water make it worse? The answer comes down to pure fire physics. Specifically, nowhere else in your home do you routinely heat a flammable liquid to within a few dozen degrees of its autoignition point. Moreover, you do it in an oxygen-rich room while distracted. This post walks through the numbers, the physics, and the fixes.
Kitchen fire statistics are worse than you think
The NFPA’s 2023 Home Cooking Fires report found that U.S. fire departments respond to an average of 158,400 home cooking fires per year. These incidents kill 470 civilians, injure 4,150, and cause $1.15 billion in direct damage. In short, cooking leads every other cause of reported home fires and fire injuries.
For example, apartments fare far worse than houses. Cooking causes 72% of apartment fires, but only 37% of fires in single-family homes. Similarly, the UK Home Office’s fire statistics for England recorded 22,877 accidental dwelling fires in the year ending March 2025. Cooking appliances sparked 44% of them. Meanwhile, the London Fire Brigade puts the share of fires starting in the kitchen at around 60%.
France sees roughly 277,000 domestic fires a year. According to the Ministère de l’Intérieur, one in five starts in the kitchen.
Temporal patterns reveal the mechanism. Fires cluster between 5 p.m. and 8 p.m., right in the middle of dinner prep. Thanksgiving tops every other day of the year for kitchen fire risk, with nearly four times the daily average. Similarly, Christmas Day, Christmas Eve, and Easter all spike 40–72% above average. Therefore, the common thread has nothing to do with equipment. It comes down to distraction.
NFPA also found that unattended equipment caused 28% of cooking fires and 48% of kitchen fire deaths. In addition, the U.S. Fire Administration pins it higher, at 37% of non-confined residential cooking fires. Consequently, stepping away from the stove ranks as the single riskiest thing a cook does.
The physics of a pan that wants to kill you
A kitchen fire differs from a miniature campfire. Instead, it behaves as a pre-loaded flammable-liquid pool fire sitting a few degrees below its own autoignition temperature. Four temperatures matter here, and people routinely confuse them.
First, the smoke point is when the oil starts shedding volatile decomposition products. Next, the flash point is where the vapours will ignite briefly if hit by a spark. Then, the fire point is where combustion becomes self-sustaining. Finally, the autoignition temperature is where the oil ignites on its own, with no spark needed.
The canonical values come from the Institute of Shortening and Edible Oils. Canola oil smokes at 236 °C, flashes at 326 °C, and reaches fire point at 350 °C. Similarly, soybean, peanut, corn, and sunflower oils cluster in the 319–334 °C flash range. Moreover, a peer-reviewed study by Liu et al. (2004) measured an autoignition temperature of 368 °C for commercial vegetable oil. Once ignited, the burning surface climbs to about 400 °C.
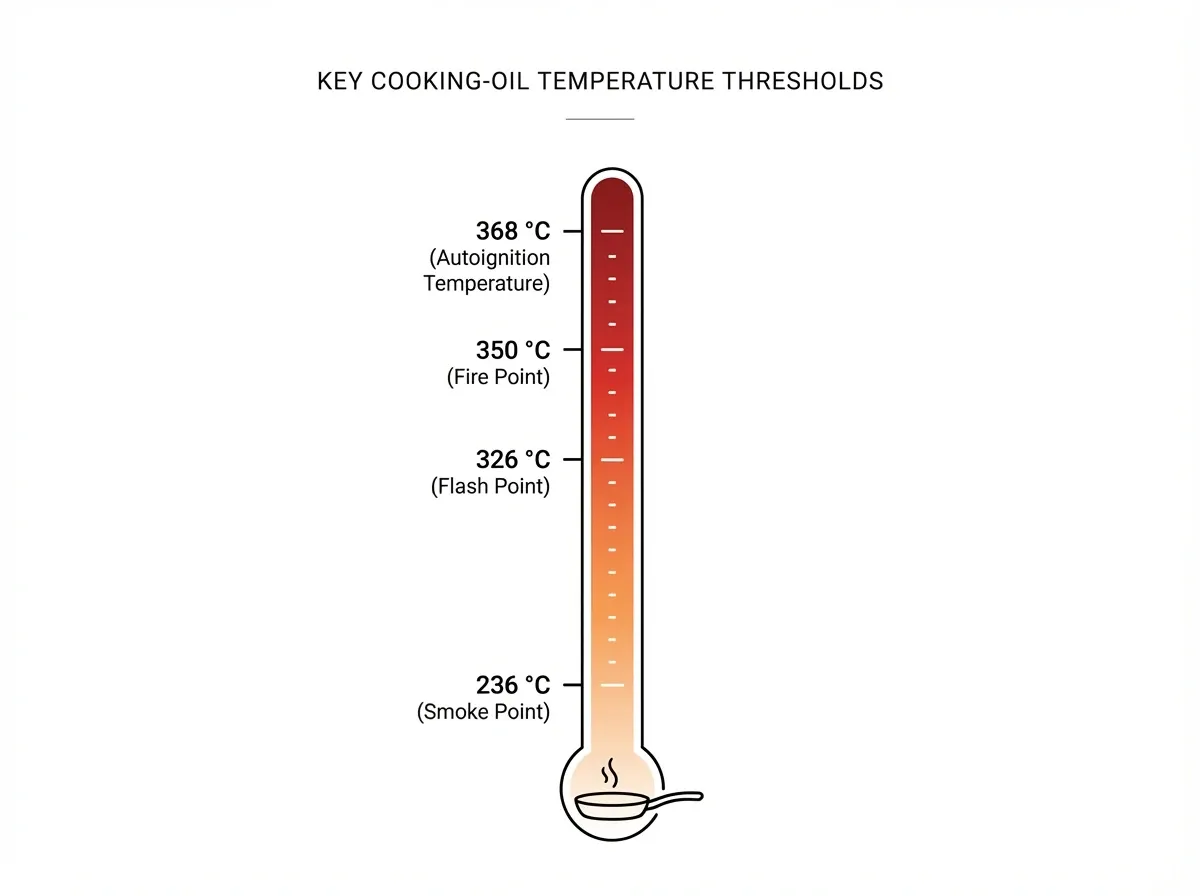
The takeaway cuts to the chase. When you deep-fry at 180 °C, you are sitting at roughly half the autoignition temperature. Therefore, forget the pan for ten minutes and you will close the remaining 180 °C gap while answering the doorbell. For the full picture of how combustion sustains itself, see our science of fire post.
Why water makes a kitchen fire catastrophically worse
Every chemistry student learns that water expands into steam by a factor of 1,700 at 100 °C. However, very few people know what that number becomes on top of oil at 370 °C. Specifically, it climbs to roughly 2,500 to 2,700 in the first fraction of a second. Therefore, a single cup (250 mL) of water tipped into a burning pan generates roughly 600 litres of superheated steam, essentially instantly.
Geometry makes the outcome worse. Water at 20 °C has a density near 1,000 kg/m³. By contrast, hot oil at 300 °C is closer to 800 kg/m³. As a result, the water sinks through the burning oil before it can boil. Once the water-oil interface hits roughly 120 °C, nucleate boiling starts from below. Fire-safety researchers call the phenomenon thin-layer boilover.
The eruption then lofts micron-scale droplets of burning oil into an aerosol fireball. Moreover, the radiative flux can reach 2 to 10 times the original steady-state pool fire, per Ferrero et al. (2007). The foundational physics comes from crude-oil pool fires, but the trigger — a water sublayer hitting 120 °C — transfers directly to cooking oil. For deeper context on how oils decompose before they ignite, see our post on the science of pyrolysis.
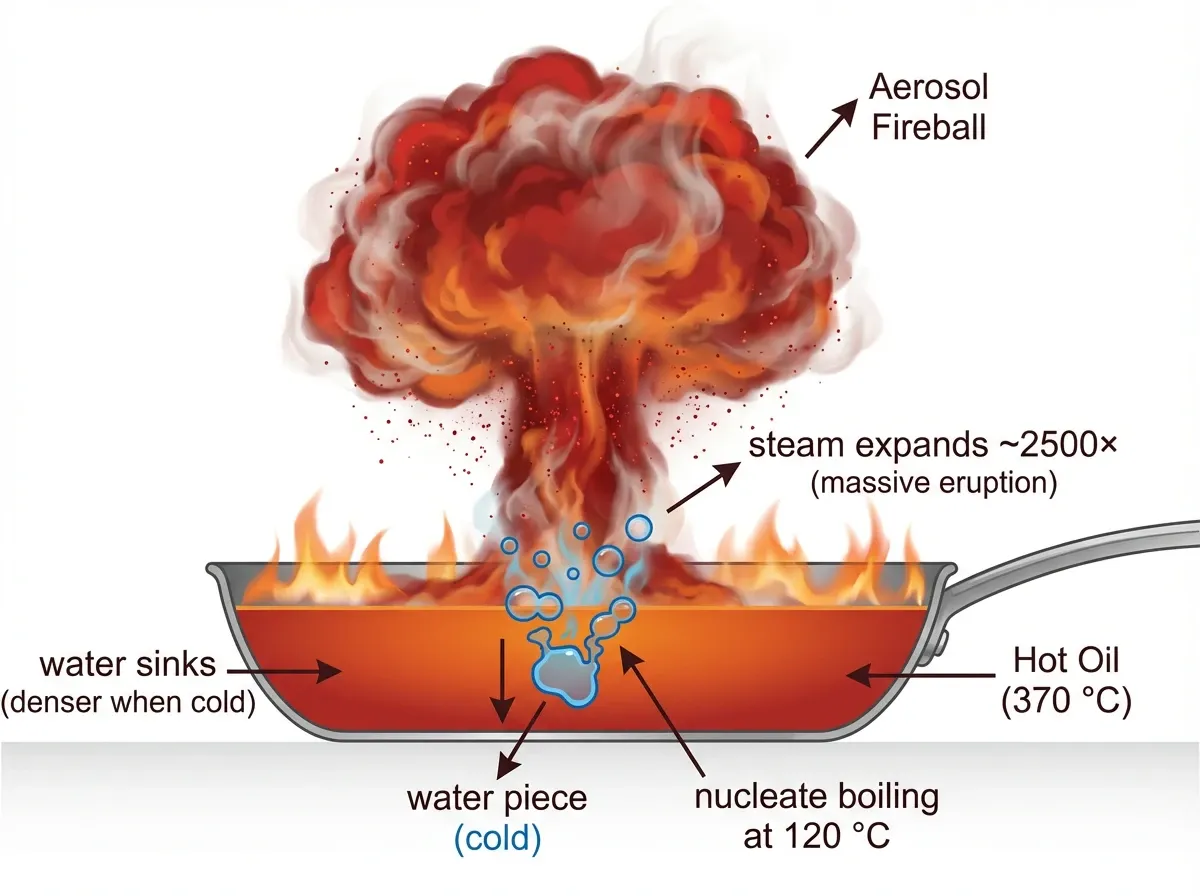
This is why the French Sapeurs-Pompiers estimate that more than 35% of serious domestic burns in France come from mishandled cooking fires. Similarly, the London Fire Brigade, the NFPA, and every fire service in the industrialised world have a single unanimous message. Never, under any circumstances, pour water on a grease fire.
What actually works, ranked by science
The correct response to a kitchen fire follows a clear hierarchy. Each step targets a different side of the fire tetrahedron: fuel, oxidiser, heat, and chain reaction.
First, the lid. Sliding a metal lid or cookie sheet over the pan starves the fire of oxygen. Then, turn off the burner. Most hydrocarbon flames self-extinguish below roughly 14–15% oxygen (Drysdale, An Introduction to Fire Dynamics, 3rd ed., Wiley, 2011). Leave the lid in place until the pan cools completely. Otherwise, reflash — the re-ignition that follows — becomes the most common failure mode.
Second, a fire blanket. Modern blankets certified to BS EN 1869:2019 now face a Class B heptane test and must insulate against 1,000 V. Moreover, certification now covers pans holding up to 3 L of cooking oil. The 2019 standard also made blankets single-use after deployment. Mount yours 1–2 metres from the cooker. In short, close enough to grab, but far enough that the fire cannot block access.
Third, a wet-chemical extinguisher. In the United States the rating is Class K. In Europe and the UK, the same class goes by Class F. Wet-chemical agents use potassium acetate, citrate, or carbonate. Consequently, they do three things at once. First, they exploit the latent heat of vaporisation of water (2,257 kJ/kg) to cool the oil below its autoignition point. Second, they displace oxygen with steam. Finally, they saponify the hot triglyceride fuel into a thick potassium soap that physically seals the oil surface.
This saponification reaction matches the exact chemistry that UL 300 has standardised for commercial kitchen suppression since November 1994. The alkaline potassium salt reacts with the triglyceride to form soap and glycerol. As a result, the foam layer prevents vapour release and blocks re-admission of oxygen.
Fourth — and often harmful — the standard ABC dry-chemical extinguisher. The household kind uses monoammonium phosphate. On a cooking-oil fire, however, it fails three ways at once. First, it offers almost no cooling, so the oil stays at 370 °C and re-ignites as soon as the powder settles. Next, its high-pressure discharge splatters burning oil out of the pan. Finally, it does not saponify. NFPA 10 therefore explicitly warns against using ABC dry chemical on a kitchen fire involving cooking oil.
A final household-remedy note on kitchen fire response. Baking soda can work on very small, incipient fires because it decomposes above 80 °C and releases CO₂. However, never dump it on a deep fryer — the rapid gas release splatters hot oil. Likewise, never throw flour. Finely divided flour has a minimum explosible concentration near 50 g/m³. Therefore, tossing it toward an open flame creates a dust-cloud deflagration — a fireball, not a smother.
The smoke alarm paradox
Working smoke alarms cut the risk of dying in a home fire by roughly 60%. Yet nearly three of every five U.S. home-fire deaths happen in homes without an alarm, or with a non-working one. The leading reason for disconnection is nuisance alarms from cooking. Specifically, 73% of people who had disabled an alarm blamed cooking in a 2010 NFPA/Harris survey.
The physics explains the paradox. Ionisation alarms trigger on small combustion particles, so cooking vapours set them off constantly. Photoelectric alarms, by contrast, respond to larger smoke particles and are far less reactive to cooking. Consequently, NFPA 72 requires alarms to sit at least 10 feet (3.0 m) from a fixed cooking appliance. Moreover, from January 2025, every alarm installed within 20 feet of a cooking appliance must pass the UL 217 8th-edition cooking-nuisance test.
France took a different path. Since the Loi Morange of 9 March 2010, every French dwelling must have a photoelectric-only detector. Notably, French law bans ionisation detectors outright. Since the rule took effect in 2015, French domestic fire deaths have dropped from roughly 800 per year to around 200.
The 90 seconds that decide everything
The most important behavioural finding of the last decade concerns how quickly modern homes actually burn. For example, UL’s Fire Safety Research Institute has shown that rooms furnished with synthetic materials flash over in under three to five minutes. By contrast, rooms furnished with natural materials of 50 years ago took roughly 30 minutes. Therefore, your grandmother’s kitchen fire gave her a quarter of an hour. Yours may give you ninety seconds.
This collapses the decision window. Fight the fire only when every one of these conditions holds: the flame stays small, contained, and steady; everyone else has left the room; emergency services have your call; and you have a clear path of retreat. Otherwise, get out. Then close the door behind you. UL’s “Close Before You Doze” experiments found that rooms with closed doors stayed below 100 °F while open-door rooms exceeded 1,000 °F.
Call emergency services first — 911 in the U.S., 999 in the UK, 112 across the EU. In addition, never move a burning pan. Both the London Fire Brigade and Lancashire Fire and Rescue identify pan-carrying as a leading source of civilian burn injuries. Finally, if you must leave the kitchen for even thirty seconds, turn the burner off.
Key takeaways
Your kitchen ranks as the most dangerous room in your house for a simple reason. You routinely heat oil to within 30–50 °C of its autoignition point, while doing a cognitively demanding task in a distracting environment. Meanwhile, modern synthetic furnishings convert a contained pan fire into a fully involved room fire in under three minutes.
The practical implications stay narrow, yet non-negotiable. First, install a photoelectric smoke alarm at least 3 metres from the stove, plus a heat detector inside the kitchen itself. Second, mount a BS EN 1869-certified fire blanket within 1–2 metres of the cooker. Third, keep a lid that fits your largest pan within arm’s reach. Fourth, if you own an extinguisher, make it Class K (U.S.) or Class F (Europe) — not ABC.
Finally, remember the hierarchy when something goes wrong. Turn off the heat. Smother with a lid or blanket. Close the door. Call the fire service. Get out. In short, water does more than merely fail on a kitchen fire. It ranks as the single most common way a contained pan fire becomes a fatal house fire. The deadliest mistake in the kitchen is the one your reflexes will tell you to make.
Cite this article
Dinh, D. C. (2026, April 19). The Science of a Kitchen Fire: Why Water Makes It Worse. PyroRisk. https://pyrorisk.net/blog/kitchen-fire-science/
Recent posts
Featured posts
Categories
Comments
Related posts

Why a LiPo Battery Is Really a Lithium-Ion Battery
A LiPo battery is a lithium-ion battery. The polymer only names the gel and pouch. Here is why that matters for fire safety.

The Science of Fire: What You Think You Know Is Wrong
Explore the science of fire — from radical chain reactions and flame anatomy to flashover physics and why modern homes burn 7x faster than 1950s ones.

What Is Fire? The Simple, Complete Guide Anyone Can Follow
What is fire? A beginner-friendly guide to combustion, the fire triangle, flame colors, fire classes, and essential home fire safety.