The Science of Fire: What You Think You Know Is Wrong
Explore the science of fire — from radical chain reactions and flame anatomy to flashover physics and why modern homes burn 7x faster than 1950s ones.

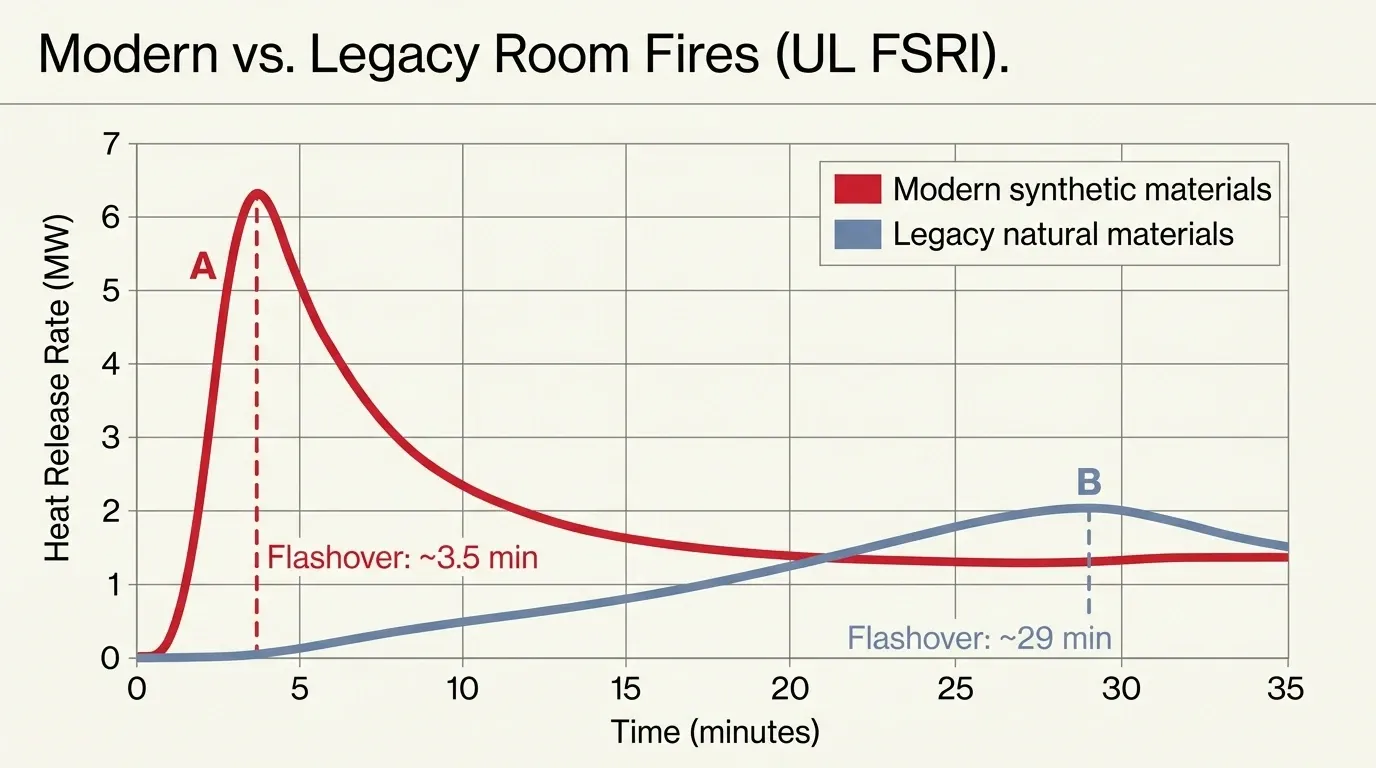
A modern living room can reach flashover in under four minutes. That means everything in the room ignites at once. A nearly identical room with 1950s-era furniture? Twenty-nine minutes. Most people have never heard of this seven-fold gap, yet it stands as one of the most important findings in the science of fire according to UL FSRI’s side-by-side burn research and Kerber’s analysis in Fire Technology.
Every day, we watch candles flicker, grill over open flame, and warm ourselves by fireplaces. Yet almost none of us could describe what is actually happening. In other words, fire remains the most familiar yet most misunderstood phenomenon in everyday life.
This post begins something I’ve wanted to build for a long time: a place to explain the science of fire with rigor, clarity, and respect. After all, fire kills roughly 3,500 Americans per year and causes $19 billion in direct property damage. Moreover, it shapes the design of every building you walk into. The science behind it turns out to be elegant, surprising, and urgent. So let’s start at the beginning.
Fire is a process, not a thing
Here is the first misconception to discard: fire is not a substance. It is not a solid, liquid, gas, or plasma — although the hottest parts of intense flames do weakly ionize. Rather, fire is a chemical process. More precisely, it is rapid exothermic oxidation that releases enough energy to sustain itself and produce visible light. In other words, the luminous, flickering thing we see amounts to hot gas undergoing violent chemistry.
From phlogiston to oxygen
For over a century, however, scientists got this wrong. The dominant theory from the 1660s through the 1770s was phlogiston. According to Johann Joachim Becher and Georg Ernst Stahl, all combustible materials held an invisible fire-substance called phlogiston. Burning released it. Overall, the theory seemed elegant. For instance, it explained why things stopped burning in closed containers — the air became “saturated” with phlogiston. It also explained why charcoal could restore metals from their ores.
However, there was one devastating problem. When metals burned, the resulting oxide weighed more than the original metal. If phlogiston escaped, the product should weigh less. Despite this, some defenders proposed that phlogiston had negative weight. Unsurprisingly, even 18th-century scientists found that hard to swallow.
Eventually, Antoine-Laurent Lavoisier killed phlogiston in the 1770s and 1780s. Using sealed vessels and precision balances, he showed that combustion was not a release but a combination. Specifically, fuel combined with a newly identified element he named oxygen. The weight gain then made perfect sense: the metal absorbed oxygen from the air. In one stroke, Lavoisier established the oxygen theory of combustion and conservation of mass. As a result, he founded modern chemistry. Tragically, he was guillotined during the French Revolution in 1794.
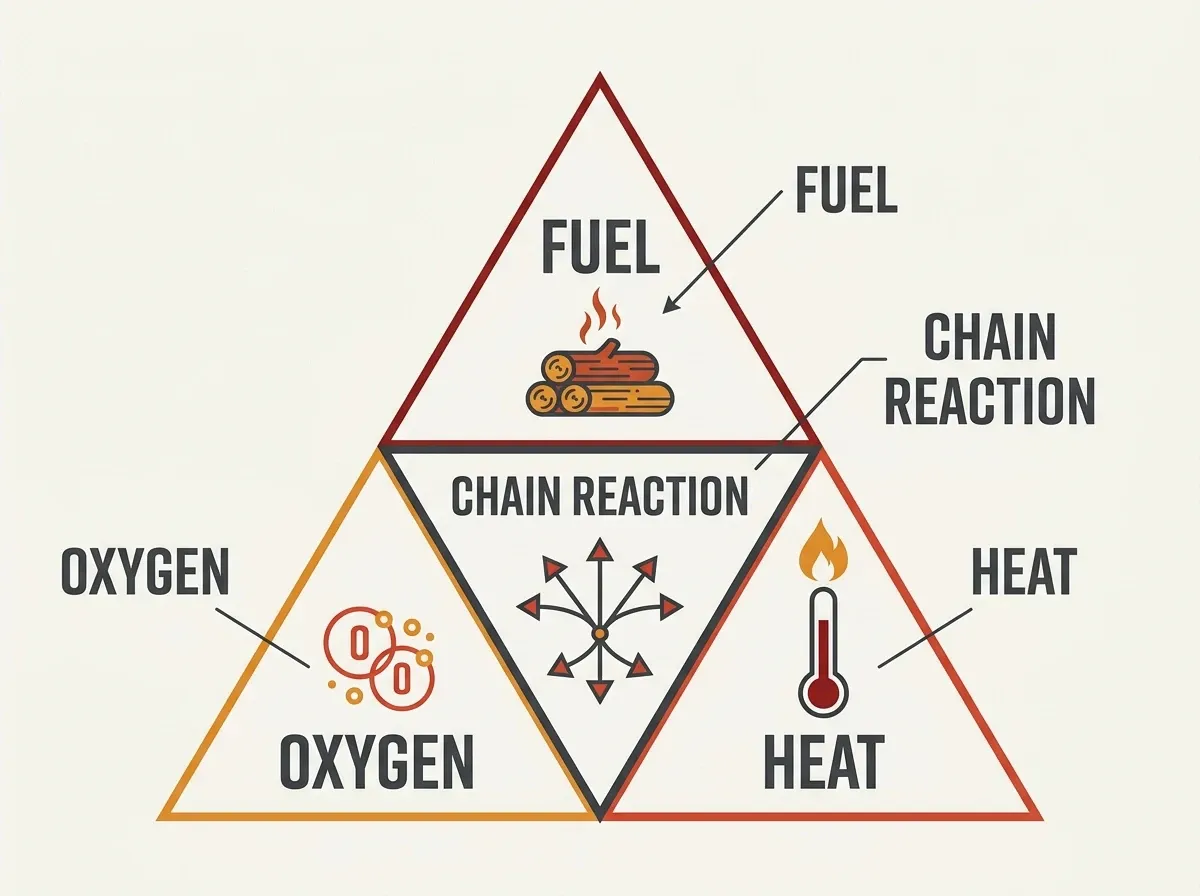
The fire tetrahedron
Today, the science of fire rests on the fire tetrahedron: fuel, oxygen, heat, and an uninhibited chemical chain reaction. Its first three faces form the classic “fire triangle.” Although necessary, they are not sufficient on their own. Scientists added a fourth element — the chain reaction — because certain agents (halons, dry chemical powders) suppress fire without removing heat, fuel, or oxygen. Instead, these agents chemically interrupt the radical chain reactions that sustain combustion. Consequently, remove any face of the tetrahedron and the fire dies.
The radical chain reactions at the heart of the science of fire
At its core, combustion is an oxidation reaction. Fuel molecules surrender electrons to oxygen, and the energy difference between reactant bonds and product bonds escapes as heat. To illustrate, the complete combustion of methane looks simple on paper:
However, that tidy equation hides a storm of intermediate chemistry.
How radicals multiply
In reality, the mechanism involves dozens to hundreds of reactions driven by free radicals. These are molecular fragments with unpaired electrons, and they react ferociously. The key players are H•, OH•, and O• radicals. Here is the critical sequence: first, a hydrogen radical attacks an oxygen molecule, producing OH• and O•. Then, that oxygen atom attacks another fuel molecule. It generates another OH• and regenerates the original H•. One radical becomes three. This exponential growth — called chain branching — makes combustion explosive rather than merely warm.
Whether a mixture ignites or merely smolders depends on a competition between two forces. On one hand, chain branching multiplies radicals. On the other hand, chain termination destroys them at surfaces or through three-body collisions. Ultimately, Nikolay Semyonov and Cyril Hinshelwood worked out this framework, earning the 1956 Nobel Prize in Chemistry for one of the most elegant theories in physical chemistry.
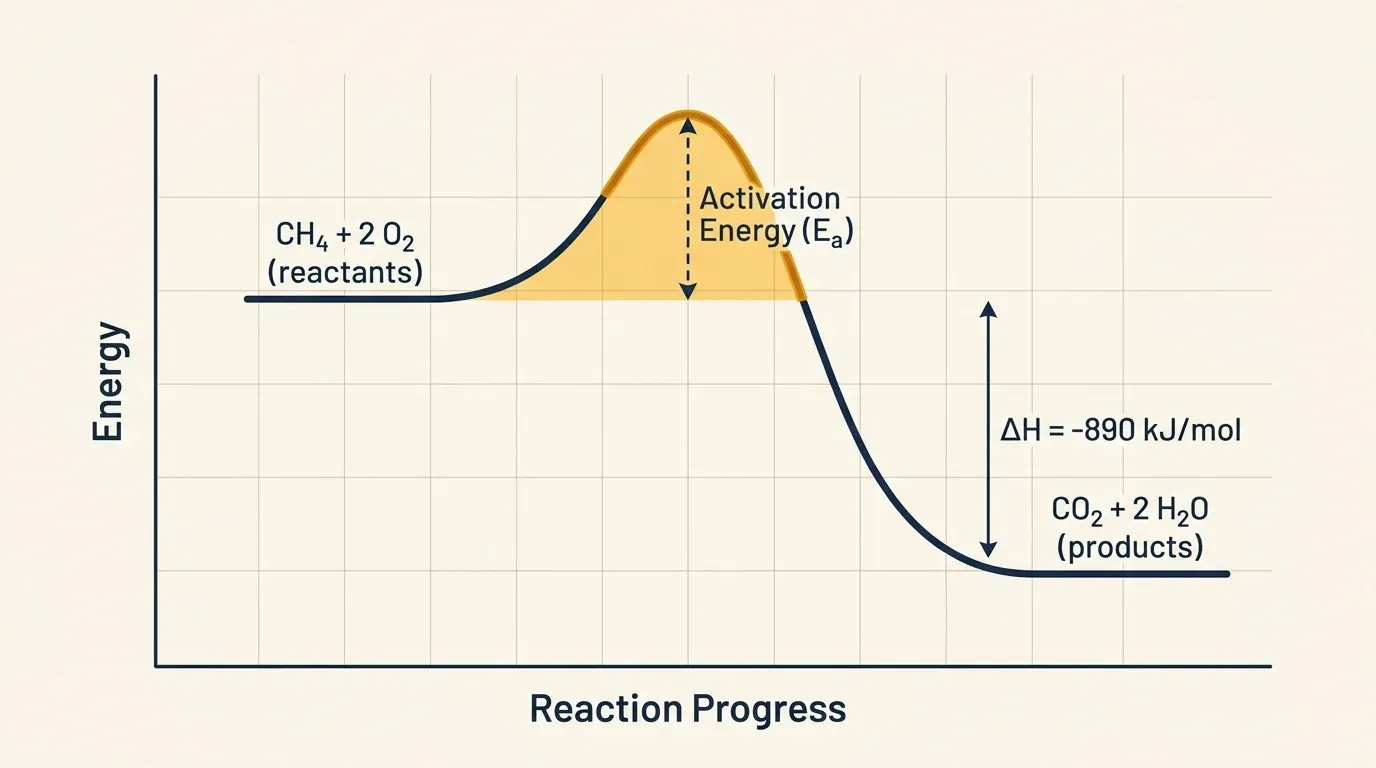
Why fires need a spark
This chain reaction machinery also explains why fires need ignition. Essentially, an initial energy input must break the first bonds and generate the first radicals. Scientists call this barrier the activation energy. Even though there is a huge thermodynamic drive toward combustion, the quantum mechanical rules for molecular oxygen create a kinetic barrier. Specifically, O₂ in its ground state has two unpaired electrons in a triplet configuration. As a result, direct reaction with singlet-state fuel molecules is a “forbidden transition.” Therefore, something — a match, a spark, or enough heat — must force the system over that barrier.
Once lit, however, the flame sustains itself. Exothermic reactions create heat, which in turn creates new radicals, which drive more reactions. In essence, it is a self-reinforcing loop that continues until fuel runs out or the chain breaks.
Incomplete combustion and its deadly products
When oxygen supply is restricted, incomplete combustion produces carbon monoxide and soot instead of CO₂. Critically, CO binds to hemoglobin roughly 250 times more strongly than oxygen. That is why smoke inhalation — not burns — is the leading cause of fire death. Furthermore, the yellow glow of a campfire signals this incomplete combustion, coming from incandescent soot particles radiating thermal energy. A perfectly combusting flame, by contrast, burns blue. This chemistry is central to how the science of fire explains real-world hazards.
Anatomy of a candle flame — the science of fire in miniature
Michael Faraday’s 1860 lectures, The Chemical History of a Candle, remain among the finest popular science talks ever delivered. He understood that a single candle holds nearly all of the science of fire in miniature. Essentially, a steadily burning candle is a laminar diffusion flame. Specifically, fuel vapor from melted wax travels upward by capillary action. It then diffuses outward while oxygen diffuses inward. When they meet, they react at an interface that reaches over 1,400°C. The result: a structured system with four distinct zones.
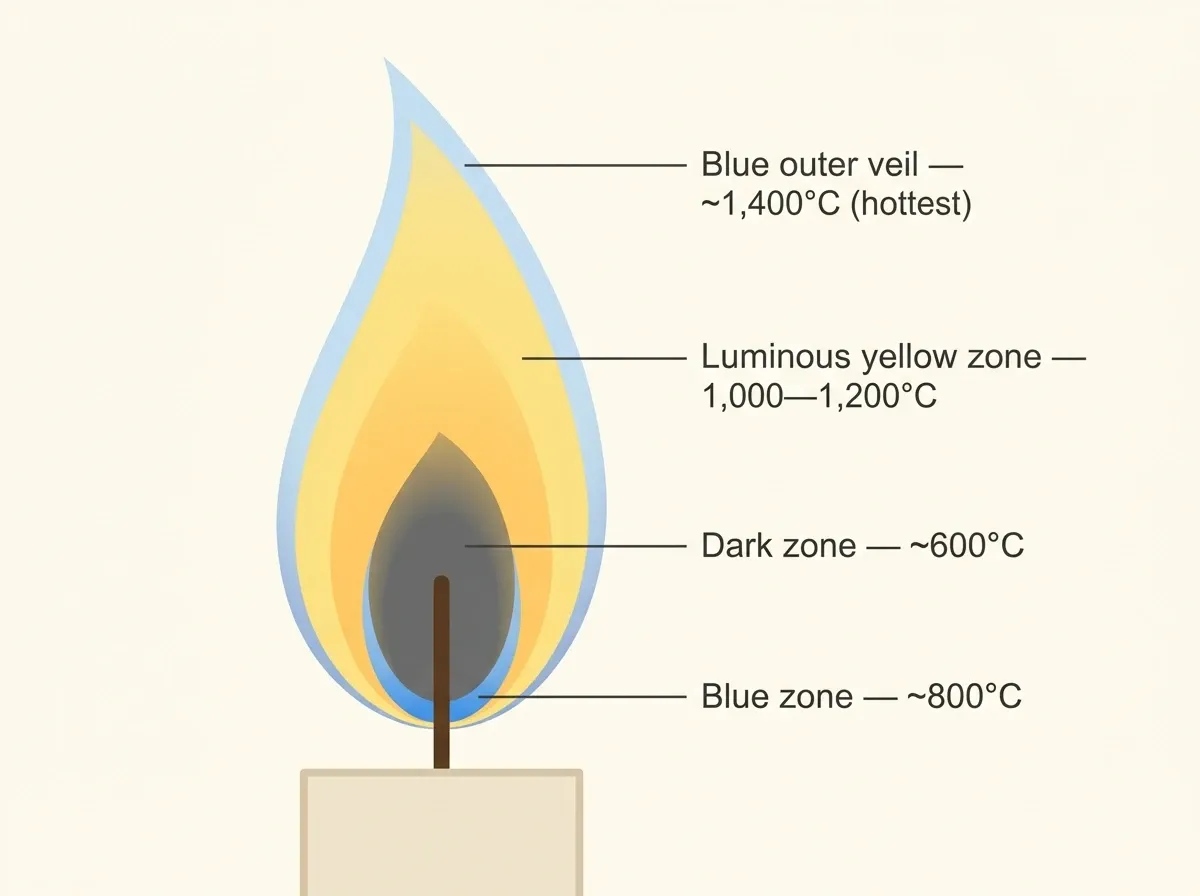
Four zones of a candle
At the center, the dark zone surrounds the wick, holding hot wax vapor with no oxygen at roughly 600°C. Because there is no oxygen, no combustion happens here. Below that, at the base, a thin blue zone marks where hydrogen first meets oxygen and burns cleanly. The blue color comes from excited CH• and C₂• radicals emitting at wavelengths around 430 and 516 nm.
Above that sits the dominant luminous yellow zone. In this region, oxygen falls short for complete combustion, so carbon fragments clump into soot particles 10–100 nm across. When heated to roughly 1,200°C, these particles glow by blackbody radiation — the same physics as a lightbulb filament.
Finally, a faint blue outer veil wraps the entire flame where it contacts air. Surprisingly, this nearly invisible zone is the hottest part, reaching about 1,400°C according to the National Candle Association. In other words, the brightest region of the flame is not the hottest. Indeed, that fact surprises almost everyone.
Premixed vs. diffusion flames
Understanding the distinction between premixed and diffusion flames is essential to the science of fire. When you open the air port on a Bunsen burner, fuel mixes with air before combustion. The result is a clean, hot blue flame. Close the port, however, and you get a sooty yellow diffusion flame. Nearly all unwanted fires — structure fires, wildfires, vehicle fires — burn as diffusion flames, because fuel and air simply do not premix by design.
What flame color tells us
Flame color also reveals chemistry. For example, blue comes from radical emission in clean combustion, while yellow and orange come from hot soot glowing by blackbody radiation. In addition, metal salts produce the vivid hues of fireworks. Sodium emits intense yellow at 589 nm — so potent that even a trace turns any flame yellow. Similarly, copper gives green, strontium gives red, and barium yields apple green according to flame test spectroscopy. In short, each element produces a unique spectral fingerprint.
Fire without gravity
Perhaps the most striking example of how gravity shapes fire comes from NASA’s microgravity experiments on the International Space Station. Without buoyancy, candle flames in space become spherical and blue — small, dim, and soot-free. This happens because oxygen reaches the flame only by molecular diffusion, which spreads equally in all directions. Consequently, the iconic teardrop shape of flames on Earth exists only because hot gas rises. Moreover, in 2012, ISS researchers found something even more remarkable: cool flames, an invisible mode of low-temperature combustion never before observed.
How fires kill: the three modes of heat transfer
Understanding heat transfer is central to the science of fire. Heat moves through three mechanisms, and each one dominates at a different stage. Together, they explain why fires spread the way they do.
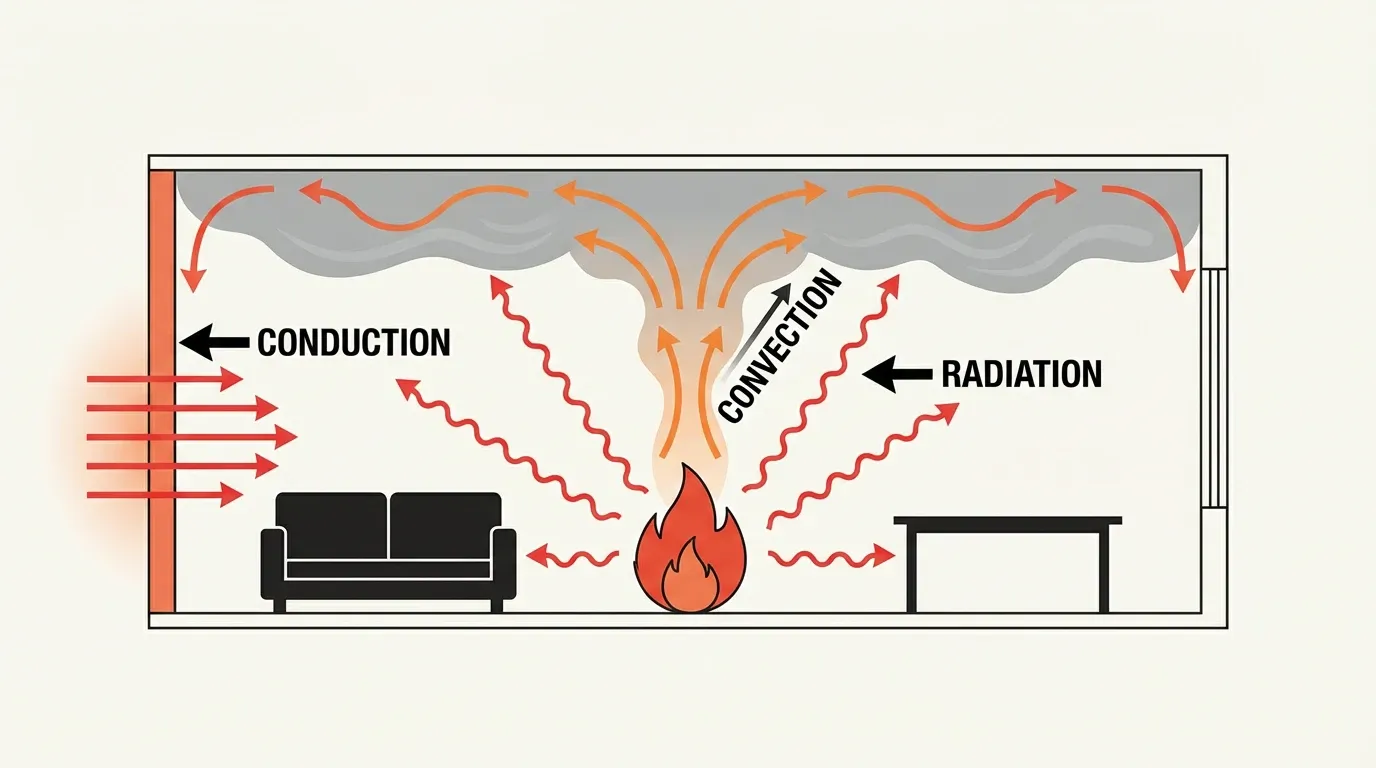
Conduction
Conduction moves heat through direct molecular contact. It plays a supporting role, mostly preheating solid fuels and transferring heat through structural elements. For example, metal fasteners, beams, and pipes can conduct heat through walls and floors, which can then ignite materials in concealed spaces. Nevertheless, in open fires, conduction contributes the least of the three mechanisms.
Convection
In the science of fire, convection drives the early stages of growth. As hot combustion gases rise, they form a fire plume that pulls in ambient air while climbing. When the plume hits a ceiling, it then spreads radially as a thin, fast-moving ceiling jet. Importantly, this jet triggers sprinkler heads and smoke detectors.
More importantly, convection carries lethal smoke and toxic gases through buildings far faster than flames travel. Because of this, smoke inhalation kills people in rooms the fire has not yet reached. Below about 400°C, convection dominates heat transfer from the fire to its surroundings.
Radiation
Radiation, by contrast, takes over as the fire grows. The Stefan-Boltzmann law governs this process, and radiative heat transfer scales with the fourth power of temperature:
As a result, a small increase in flame temperature produces an enormous increase in radiated energy. In large fires, radiation accounts for 80–90% of total heat flux. Even more critically, radiation creates devastating positive feedback loops. First, flames radiate heat to surrounding fuel. Next, that fuel pyrolyzes and produces combustible vapor. Then, the vapor feeds larger flames, which radiate still more heat. Meanwhile, the hot smoke layer at the ceiling radiates downward onto every exposed surface. Ultimately, this feedback loop drives flashover.
Heat release rate and flashover: the science of fire meets real danger
If one number defines a fire’s danger, it is the Heat Release Rate (HRR). HRR measures how fast a fire converts chemical energy into heat, in kilowatts. In a landmark 1992 paper, NIST researchers Vytenis Babrauskas and Richard Peacock called HRR the single most important variable in fire hazard. Their reasoning was clear: although toxic gases directly cause most fire deaths, HRR drives everything else. Specifically, it controls temperature rise, smoke production, toxic gas generation, fire spread speed, and time to untenable conditions. In short, control HRR and you control the hazard.
The numbers are sobering
Consider the range: a wastepaper basket fire produces roughly 15 kW. By contrast, an upholstered armchair can reach 3,000 kW — three megawatts in under three minutes. Even more alarming, a dry Christmas tree ignited in NIST’s lab hit 4,300 kW in twenty seconds, engulfing an entire furnished room in flashover within forty seconds.
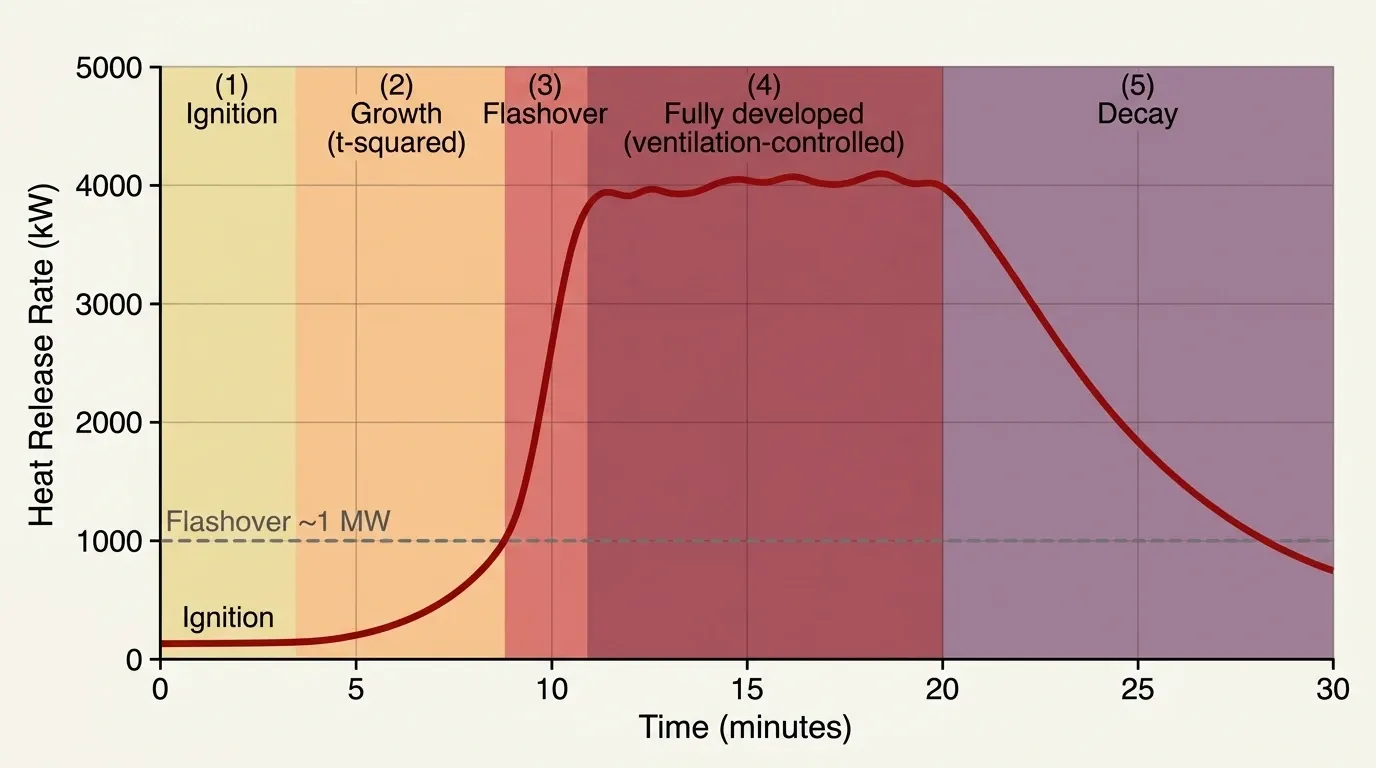
What flashover means
Flashover separates a manageable fire from a lethal one. As a fire grows, the hot upper gas layer radiates heat downward onto all exposed surfaces. When that layer reaches about 500–600°C and heat flux at floor level exceeds roughly 20 kW/m², every combustible surface in the room ignites nearly at once. Consequently, the room goes from a single burning object to total involvement in seconds. After flashover, survival time in full firefighter gear drops to about seven to ten seconds.
Before flashover, fire growth typically follows a t-squared model, where HRR grows with the square of time. Accordingly, the rate depends on fuel type: slow, medium, fast, or ultra-fast.
Fuel-controlled vs. ventilation-controlled
Whether a fire is fuel-controlled or ventilation-controlled also determines its behavior. In fuel-controlled fires, there is not enough combustible material. In ventilation-controlled fires, there is not enough oxygen. Notably, most fires that have progressed beyond early stages are ventilation-controlled by the time firefighters arrive.
This creates a terrifying paradox. Opening a door or window to a ventilation-controlled fire floods it with fresh oxygen, and the result can be an explosive HRR surge. In fact, NIST wind-driven fire experiments documented HRR jumping from 5 MW to 32 MW almost instantly when ventilation arrived. Such findings show why the science of fire matters so urgently for firefighter safety.
Modern fire science confronts an accelerating threat
Of course, the science of fire does not stay in laboratories. It shapes every building code, sprinkler specification, and fire-rated wall assembly. NIST’s Fire Dynamics Simulator (FDS), a free open-source CFD tool released in 2000, solves the Navier-Stokes equations for thermally driven flow to simulate smoke and heat transport. Today, FDS has become the industry standard for fire safety design. For instance, researchers used it to reconstruct the 2003 Station nightclub fire. In that tragedy, 100 people died in under five minutes after pyrotechnics ignited non-fire-retardant foam. The NIST simulation showed that a sprinkler system would have controlled the fire within 114 seconds.
Another key tool in the science of fire is the cone calorimeter, which Babrauskas developed at NIST in 1982. It measures HRR, time to ignition, smoke output, and toxic gas yields using oxygen consumption calorimetry. The core principle is simple: roughly 13.1 MJ of heat escapes per kilogram of oxygen consumed, regardless of fuel type. Over 300 units now operate worldwide, making it the most important bench-scale fire test instrument.
However, the challenge evolves faster than codes can keep up. Three emerging threats now define the frontier:
Lithium-ion battery fires
Lithium-ion battery fires use a mechanism alien to traditional combustion. During thermal runaway, a failing cell generates oxygen internally and fuels its own fire. As a result, smothering — the conventional approach — simply does not work. Furthermore, EV battery fires can reach 2,700°C and require roughly 2,500 gallons of water to extinguish, whereas conventional vehicle fires need only 500–1,100 gallons. Worse still, batteries can reignite hours or days later. With global lithium-ion demand expected to reach nearly 5 TWh by 2030, this problem is scaling fast.
Wildfires intensified by climate change
Wildfires represent perhaps the largest-scale challenge in the science of fire. Since 1990, fire-conducive weather has increased significantly in California. When combined with wildland-urban interface (WUI) expansion, the likelihood of extreme-impact wildfires has quadrupled. To put this in context, the WUI — where houses meet wildland vegetation — grew 12.6% globally between 1985 and 2020. Meanwhile, US wildfire costs now reach an estimated $394–893 billion annually.
The modern fuel load problem
This may be the most insidious threat, because it operates invisibly inside every home. UL’s Fire Safety Research Institute ran side-by-side burn experiments and found a stark difference. Rooms with contemporary synthetic materials — polyurethane foam, polyester fabrics, polyolefin carpets — reached flashover in under five minutes. In contrast, rooms with legacy natural materials took over twenty-nine minutes.
On top of that, modern homes feature open floor plans that eliminate compartmentalization. They also use hollow-core doors and engineered lumber that collapses faster than dimensional lumber. As a consequence, firefighters and occupants now have roughly one-fifth the escape time their predecessors did. The smoke is also far more toxic.
Where we go from here
Fire was the first chemical reaction humans ever controlled, and it may be the last one we fully understand. After all, a single candle flame involves over a thousand simultaneous reactions among dozens of species. Scale that to a compartment fire, a wildfire, or a lithium-ion thermal runaway, and the complexity becomes staggering.
What strikes me about the science of fire: where it sits. It spans chemistry, physics, fluid dynamics, materials science, human behavior, and public policy. Even so, the knowledge gaps remain consequential. For example, the Triangle Shirtwaist Factory fire of 1911 killed 146 workers — many of them teenagers — behind locked doors. It gave birth to over thirty new safety laws and eventually to OSHA itself. Likewise, the Great Fire of London in 1666 destroyed 13,200 houses and invented the building code. More recently, the Grenfell Tower fire in 2017 killed 72 people because combustible plastic cladding wrapped a concrete tower. In every case, major advances in fire safety have come from tragedy.
This site exists because the science of fire should not only become accessible after the next disaster. In future posts, we will go deeper into each topic introduced here — from the fluid mechanics of fire plumes to the materials science of fire-resistant construction, from computational fire modeling to the physics of wildfire spread. Fire is one of the most fascinating and consequential phenomena in the physical world. It deserves to be understood.
Cite this article
Dinh, D. C. (2026, April 13). The Science of Fire: What You Think You Know Is Wrong. PyroRisk. https://pyrorisk.net/blog/the-science-of-fire/
Recent posts
Featured posts
Categories
Comments
Related posts

What Is Fire? The Simple, Complete Guide Anyone Can Follow
What is fire? A beginner-friendly guide to combustion, the fire triangle, flame colors, fire classes, and essential home fire safety.

The Science of Pyrolysis: How Heat Unmakes Everything
Pyrolysis — heat breaking molecular bonds without oxygen — drives wildfires, battery thermal runaway, biochar, plastic recycling, and even coffee roasting.
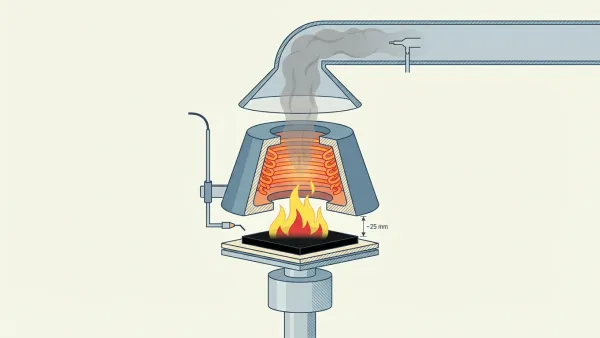
Cone Calorimeter 101: HRR, THR, and MARHE Explained
What the cone calorimeter measures — heat release rate, total heat release, and MARHE — and why each one matters for fire safety.