What Is Fire? The Simple, Complete Guide Anyone Can Follow
What is fire? A beginner-friendly guide to combustion, the fire triangle, flame colors, fire classes, and essential home fire safety.

Have you ever stared into a campfire and wondered what you were really looking at? So what is fire? It isn’t a solid, a liquid, or a gas. It isn’t even a “thing.” Fire is an event — a fast chemical reaction happening right in front of you. Every flicker tells a story of tiny fuel pieces breaking apart and locking onto oxygen from the air, giving off heat and light as they do.
Fire keeps us warm. It cooks our food. It also powered every great chapter of human history. However, fire takes thousands of lives every year and reshapes whole forests. So whether you’re a curious kid, a parent brushing up on safety, or a science-lover who just wants to understand the flames on a birthday candle, this guide walks you through it — from the tiniest molecules in a flame to the wildfires that remake a landscape.
What is fire, really?
At heart, fire is a chemical reaction called combustion. The recipe is simple. First, take a fuel (wood, wax, gasoline, natural gas). Next, mix it with oxygen from the air. Finally, add enough heat to get things started. The fuel then breaks apart and hooks onto oxygen to form new stuff — mostly carbon dioxide and water vapor. So that reaction gives off a huge amount of energy, which is why fire feels hot and looks bright.
Scientists call a reaction like this a heat-releasing reaction (the technical word is exothermic). Think of it like a ball rolling downhill. Once you give the ball a push, it keeps going on its own. Similarly, heat from the reaction breaks apart more fuel, the fuel grabs more oxygen, and the cycle keeps feeding itself.
Here’s the reaction in plain words, using natural gas as the fuel:
Methane + oxygen → carbon dioxide + water + a lot of heat and light
But at the molecular level, combustion isn’t one clean step. Instead, it’s more like a chain of tiny reactions driven by pieces of molecules called free radicals — little reactive fragments that smash into fuel and oxygen, then spawn more reactive fragments. One spark creates the first few. Those then create more. Next, the cascade grows until the whole fire is burning. For a deeper look at the chemistry, see our longer piece on the science of fire.
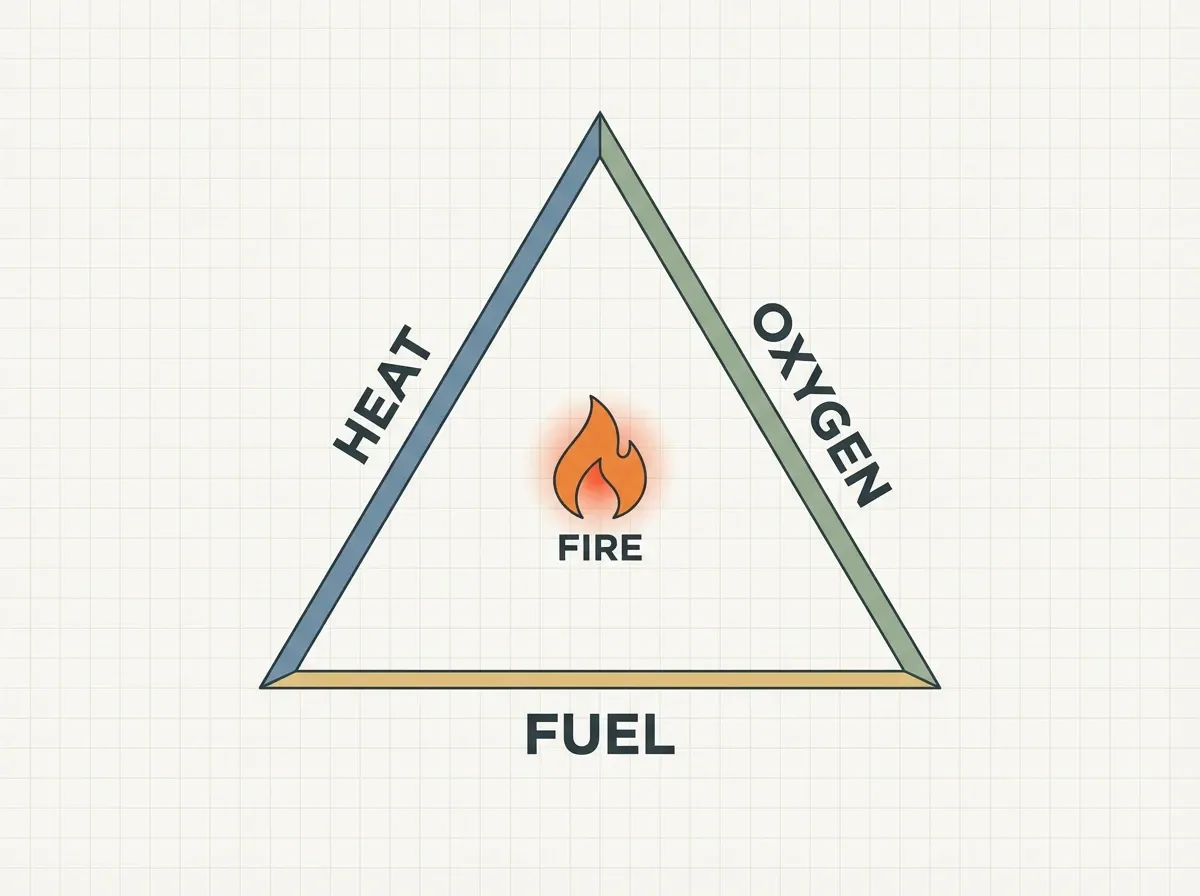
The fire triangle — and the fire tetrahedron
For a long time, firefighters and teachers have relied on the fire triangle to show what fire needs. It’s a simple picture: three sides, all needed, and if you remove any one of them, the fire dies.
The three sides are:
- Fuel — anything that can burn: wood, paper, gasoline, cloth, natural gas. First, you have to heat solid fuels until they release flammable gases. Only then do they actually burn.
- Oxygen — the air around us holds about 21% oxygen. Most fires need at least 16% oxygen to keep going. So oxygen is the ingredient that reacts with the fuel and releases the energy.
- Heat — the push that starts the reaction. A match, a spark, a lightning bolt, or even sunlight through a magnifying glass can provide it.
Modern fire science upgrades the triangle to a fire tetrahedron — a four-sided pyramid. The fourth side shows the chemical chain reaction we just talked about. Those tiny reactive fragments don’t only help fire start. They also keep it alive. Every time a fragment reacts, it makes more fragments, and so the fire feeds on itself.
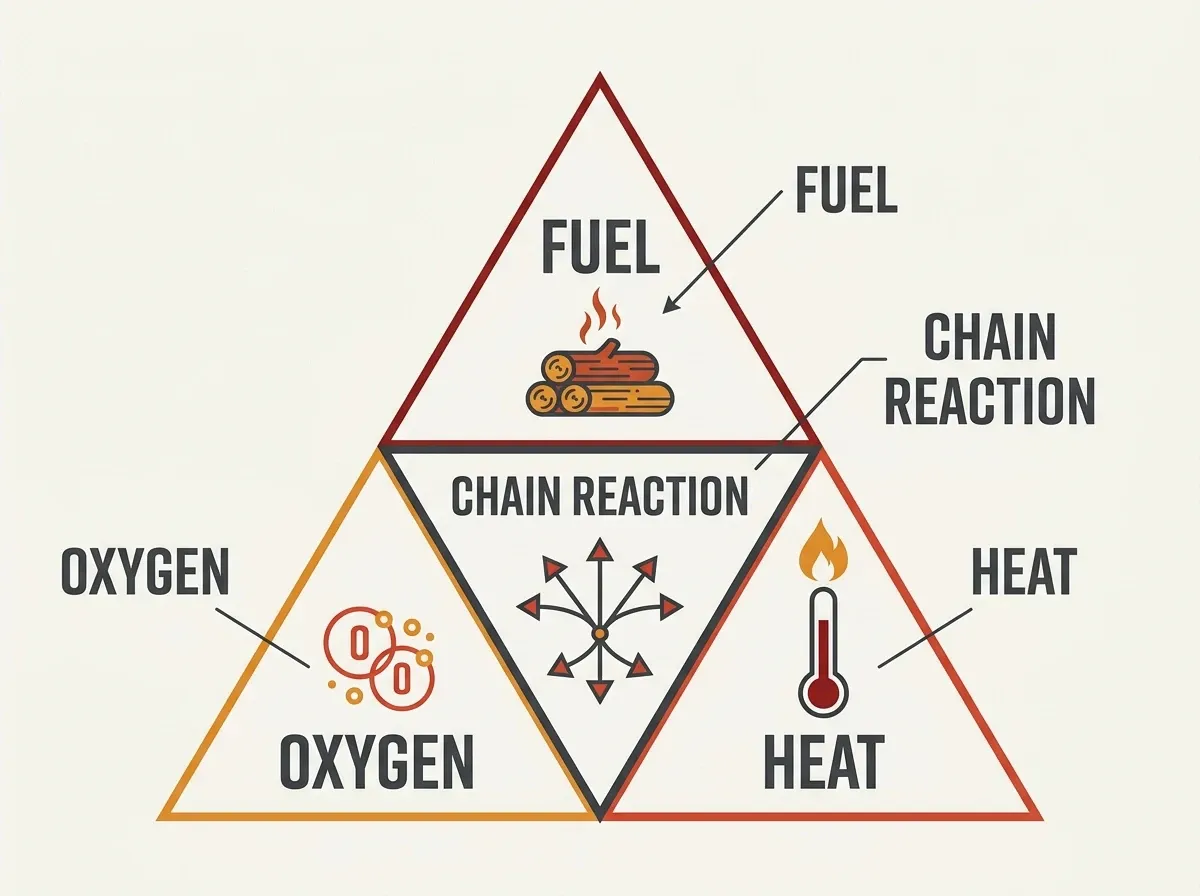
So why does the fourth side matter? Because some fire extinguishers work by breaking the chain reaction itself. Halon extinguishers, for example, release atoms that grab the reactive fragments and stop the cycle. Dry-chemical powders do something similar. The fire doesn’t run out of fuel, oxygen, or heat — instead, something simply interrupts the chemistry.
So there are four ways to put a fire out:
- Starve it (take away the fuel)
- Smother it (cut off the oxygen)
- Cool it (take away the heat, usually with water)
- Interrupt it (break the chemical chain)
Why flames come in different colors
So what is fire actually showing us when its color shifts? Not all flames look the same, and the color gives a real clue about what’s happening inside. Flames fall into two big families:
- Premixed flames happen when the fuel and air mix before they reach the flame. A gas stove burner gives a good example. These flames burn hot and clean, and they usually look blue.
- Diffusion flames happen when the fuel and air only meet at the flame itself. A candle offers the classic example. These flames burn a bit cooler, make soot, and glow yellow or orange.
Two things set the color:
- Glowing soot. Tiny soot specks inside a yellow flame get superheated and glow, just like the filament in an old light bulb. Also, the hotter the soot, the more the color shifts from dull red toward orange, yellow, and finally near-white.
- Excited molecules. In a clean-burning blue flame, reactive fragments give off light at very specific colors, which our eyes see as blue and sometimes green. Similarly, you can see this idea at work in the anatomy of a flame on Wikipedia.
Here’s a rough guide to what a flame color tells you:
| Flame color | Rough temperature | What’s happening |
|---|---|---|
| Dull red | ~700°C (1,300°F) | The coolest flame you can see |
| Orange-yellow | 1,100–1,200°C (2,000–2,200°F) | Glowing soot — incomplete burning |
| White | 1,300–1,500°C (2,400–2,700°F) | Very hot; almost complete burning |
| Blue | 1,400–1,650°C (2,550–3,000°F) | Clean burning; excited molecules |
A candle flame acts like a tiny lab for all of this. The blue ring at the bottom shows where burning runs cleanest. The bright yellow middle holds all the glowing soot. Finally, the dim outer edge shows where hot gases cool off and fade away.
How a fire grows inside a building
Fire inside a building moves heat around in three ways:
- Conduction — heat travels through objects that touch each other. For example, a metal beam heated on one side can set fire to wood touching the other side.
- Convection — heat rides on moving hot air. In an early fire, superheated air rises, pools at the ceiling, and pre-cooks everything below.
- Radiation — heat travels through open space as invisible infrared waves. So a big fire can ignite a couch across the room without ever touching it.
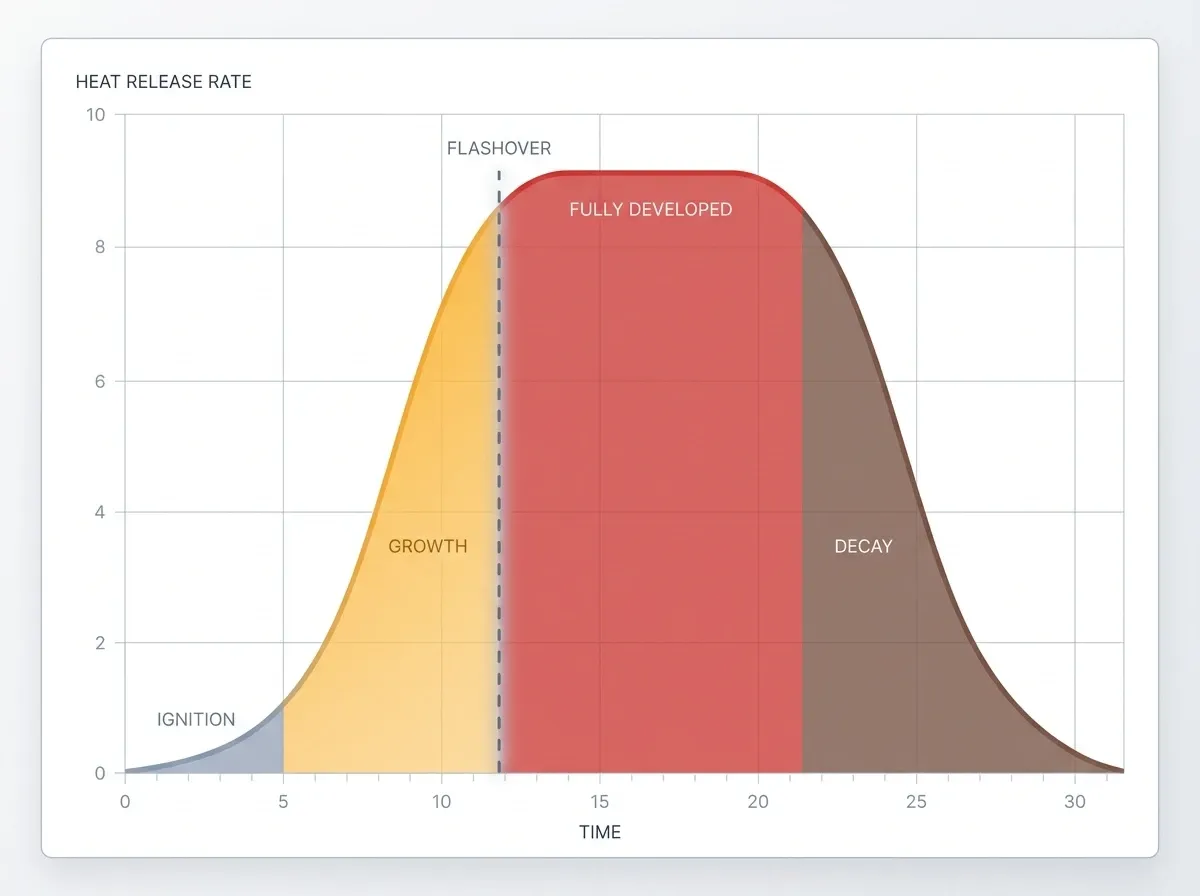
The four stages of a building fire
Knowing these stages can save your life.
1. Ignition. Heat, fuel, and oxygen meet for the first time. The fire stays small and easy to put out. So a fire extinguisher can handle it. Every second counts.
2. Growth. Now the fire feeds on nearby stuff. Hot smoke builds up at the ceiling and beams heat down onto the furniture and walls. Because of that heat, a growing fire can double in size every 60 seconds. At the end of this stage, the room stands on the edge of something far more dangerous.
3. Flashover. When the ceiling hits about 500–600°C (930–1,100°F), the radiant heat ignites everything in the room at once. Firefighters call this sudden jump flashover. It happens in a few seconds, and nobody still inside can survive it. For example, a small wastebasket fire can reach flashover in as little as three to five minutes. Read more about flashover on Wikipedia.
4. Fully developed. Every burnable surface now burns. Temperatures peak between 700°C and 1,200°C. So how much air can get in through windows and doors now sets the limit for the fire.
Decay. Finally, fuel runs low and the fire dies down. However, if the fire choked on a lack of oxygen first, suddenly opening a door can trigger a backdraft — an explosive reignition when fresh air rushes in. Firefighters train specifically for this.
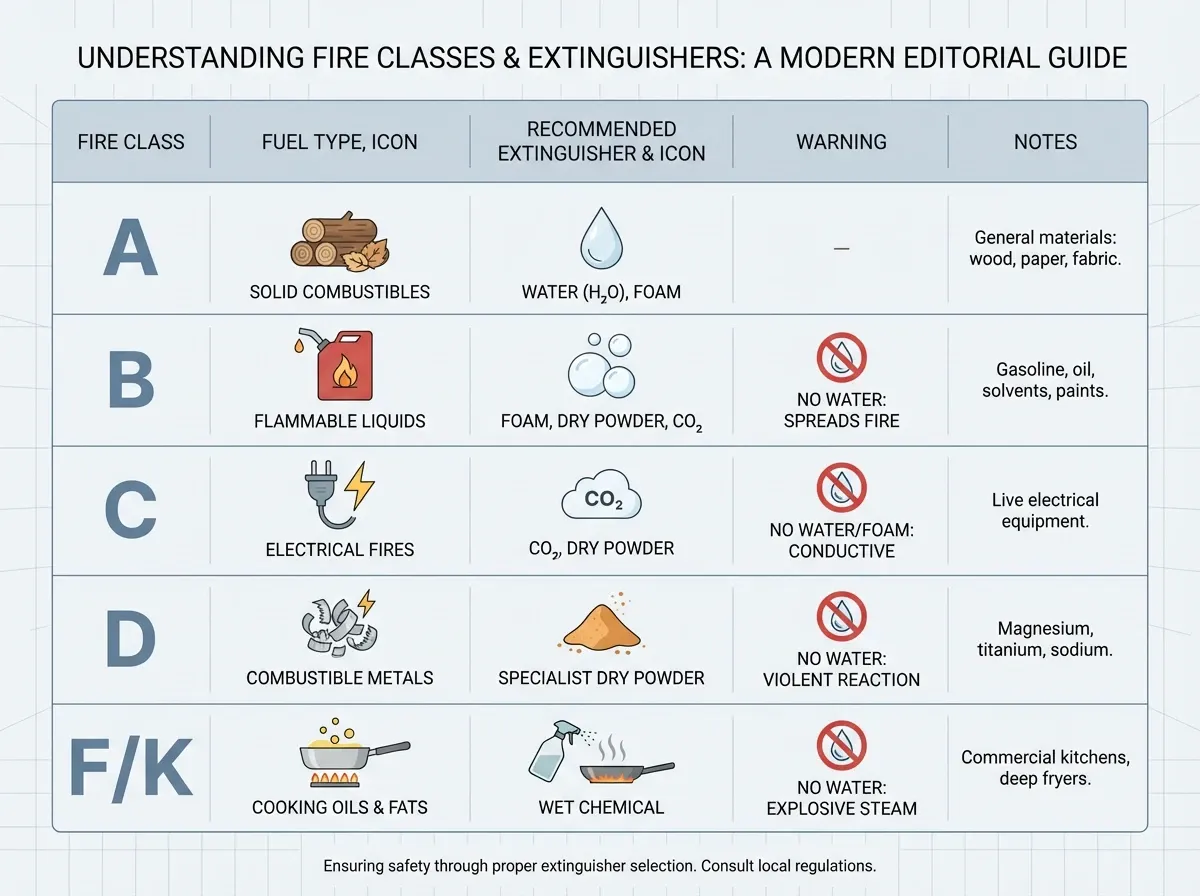
The five classes of fire
Not every fire looks the same, and using the wrong extinguisher can make things worse. So fire experts sort fires into five classes. That’s why you see letters on the front of every extinguisher.
Class A — everyday burnables. Wood, paper, cloth, rubber, and many plastics fall here. These cause the most common fires. Water works great because it cools the fuel below the temperature it needs to burn.
Class B — flammable liquids and gases. Gasoline, kerosene, alcohol, propane, and paint fall here. Never use water on these. Water can spread the burning liquid and carry flames across the room. Instead, foam extinguishers smother the fuel, and carbon dioxide (CO₂) extinguishers push the oxygen away.
Class C — electrical fires. Live equipment counts here: motors, wires, plugged-in appliances. The main danger comes from getting shocked. So use CO₂ or a dry-chemical extinguisher that doesn’t conduct electricity. Once the power goes off, the fire usually drops back to a Class A or B fire.
Class D — burning metals. Magnesium, titanium, lithium, and aluminum fall here. These burn at very high temperatures and can react violently with water. Only special dry powders (graphite, sodium chloride) work. They smother the metal by forming a crust.
Class K — cooking oils and fats. This class stands apart because modern cooking oils burn hotter than regular extinguishers can handle. A wet-chemical extinguisher cools the oil and creates a soapy layer that stops it from re-lighting. Also, for grease fires at home, Ready.gov recommends sliding a metal lid over the pan and turning off the heat.
For most homes, an ABC dry-chemical extinguisher covers the three most common fires. And there’s a classic memory trick for using one — the PASS technique from OSHA:
- P — Pull the pin
- A — Aim at the base of the flames
- S — Squeeze the handle
- S — Sweep side to side
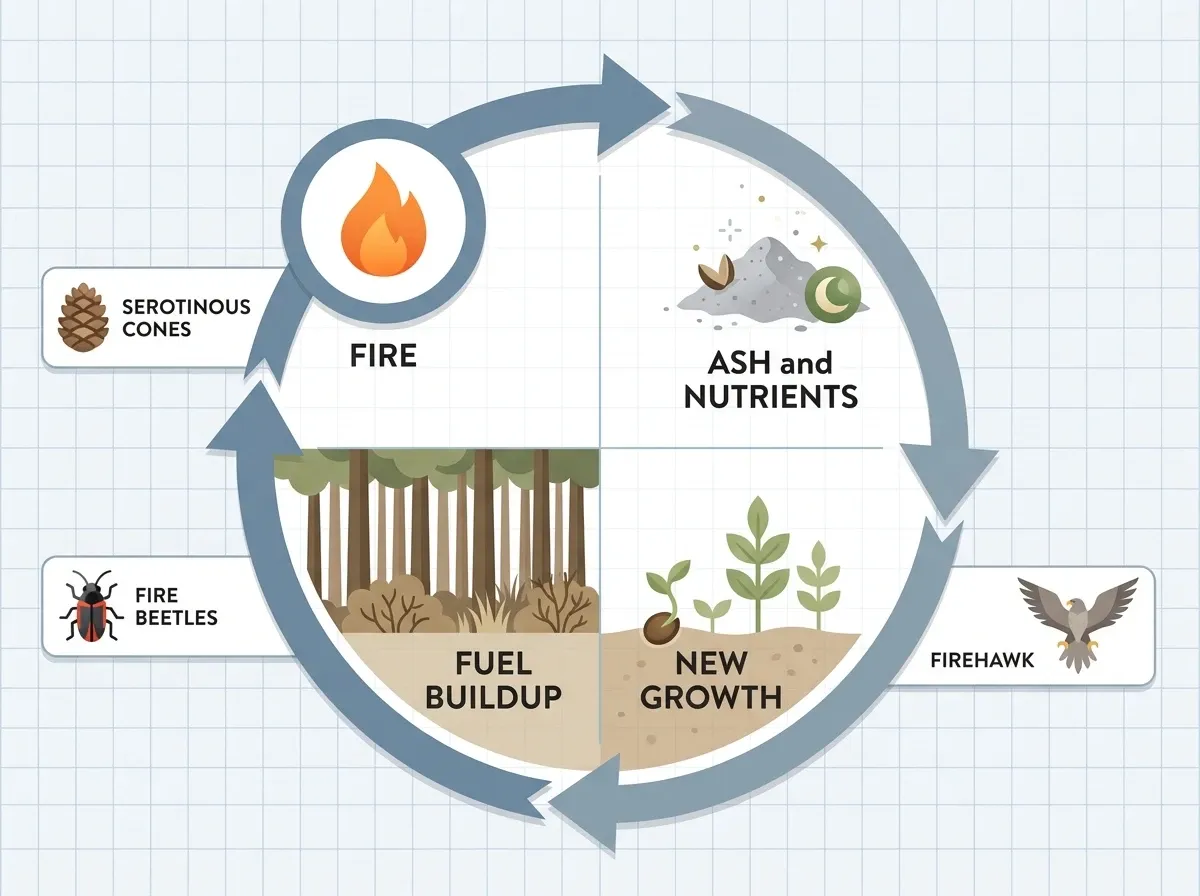
Fire in nature
Fire isn’t only destructive. In many forests and grasslands, fire plays a natural part of the life cycle. Scientists sometimes call fire a keystone process because whole ecosystems depend on it.
Wildfires: destruction and renewal
Almost 85% of wildfires in the United States come from people — unattended campfires, tossed cigarettes, sparks from equipment. On national forest land specifically, lightning tops the list of natural causes. The pattern is simple: a long, hot lightning bolt hits dry grass or a dry tree, and if conditions line up, it catches.
Meanwhile, climate change keeps making the problem worse. For example, fire seasons are getting longer and more intense, and the amount of land that burns each year has grown a lot over recent decades.
Species that need fire
Some plants and animals don’t just tolerate fire — they depend on it.
- Giant sequoia trees can’t reproduce without fire. First, they need the heat to crack open their cones. Next, they need cleared ground for baby trees. Finally, they need ash in the soil for nutrients.
- Jack pine cones hold resin that only melts above 140°C (284°F). So without a fire, the seeds never come out.
Australia gives us even stranger examples of fire-adapted life:
- Banksia plants in Australia do the same thing — their seed pods open only after fire passes through.
- Three kinds of Australian birds of prey — black kites, whistling kites, and brown falcons — pick up burning sticks and drop them in dry grass to flush out prey. Aboriginal Australians have known these “firehawks” for thousands of years, and scientists confirmed the behavior in 2017.
- The fire beetle carries heat sensors that pick up a forest fire from up to 40 km (25 miles) away. Because freshly burned trees make the safest nursery for its eggs, it flies toward the fire.
Fire safety basics everyone should know
House fires still hit homes far too often. In the U.S., a home fire breaks out roughly every 96 seconds, and cooking tops the list of causes. Also, here’s the scariest statistic: three out of five home fire deaths happen in homes with no smoke alarm, or with an alarm that wasn’t working.
Smoke alarms save lives
Working smoke alarms cut the risk of dying in a home fire by more than half. So put one in every bedroom, outside each sleeping area, and on every floor. Link them together if you can, so when one goes off, they all do. Then test them once a month, change the batteries once a year, and replace the whole alarm every 10 years.
Plan your escape — and practice it
Once a smoke alarm sounds, you may only have one or two minutes to get out. The NFPA’s home escape plan guide gives you a great place to start. First, draw a floor plan of your home and mark two exits from every room. Next, pick an outdoor meeting spot. Then, practice the plan at least twice a year (including once at night). Also, close doors behind you on the way out. A closed door can hold back flames and toxic smoke for extra minutes.
If fire catches you by surprise
- If your clothes catch fire: stop, drop, and roll. Stop moving, drop to the ground, cover your face, and roll until the flames go out.
- If the room fills with smoke: crawl low where the air stays cleaner.
- Before opening a door: feel it with the back of your hand. If it feels hot, find another way out.
- Once you get out, stay out. Never, ever go back inside a burning building for a pet or a belonging.
Fun facts about fire
Here are a few surprises — great for a school report or just for fun. First, a batch from space and chemistry:
- Fire burns as perfect spheres in space. Without gravity, hot air can’t rise, so flames on the International Space Station form round, blue orbs instead of teardrop shapes.
- Earth is the only planet we know of with fire. No other world in our solar system has enough oxygen in its atmosphere to keep combustion going.
- The Sun isn’t actually on fire. It makes its energy by fusing hydrogen into helium — nuclear fusion, not combustion. No oxygen required.
- Fire makes water. One of combustion’s main products is H₂O. For example, hold a cold spoon above a candle and you’ll see tiny water droplets collect on it.
And a few strange records from Earth:
- One fire has smoldered for over 5,000 years. A coal seam under Mount Wingen in Australia has burned underground since long before humans built the pyramids.
- A fire destroyed the fire hydrant patent. In 1836, a fire at the U.S. Patent Office burned most of its records. That included, awkwardly, the original patent for the fire hydrant.
- Fire tornadoes are real. In 2018, the Carr Fire in California produced a fire tornado with 143 mph winds and a rotating column that reached 17,000 feet into the sky.
Takeaways
So what is fire in one last sentence? Fire stands as one of the oldest forces in human life, and also one of the most misunderstood. It isn’t a thing. Instead, it’s a process — a chain of chemical reactions that release heat and light. Understanding that process lets us use fire safely and react well when it turns dangerous.
So what should you take away from this guide?
- Know the fire triangle. Take away fuel, oxygen, or heat, and the fire dies.
- Know the fire classes. The wrong extinguisher on a grease or electrical fire can make things much worse.
- Prevent more than you react. Working smoke alarms, a practiced escape plan, and basic extinguisher know-how make the difference between a close call and a tragedy.
Fire shaped the planet’s ecosystems, fueled the rise of civilization, and still powers most of modern life. So respect what fire is and what it can do — and you’ll live more safely alongside it.
Stay curious. Stay safe.
If you want to go deeper, explore more fire fundamentals on the site.
Further reading
For anyone who wants to dig further, here are some trustworthy sources used in this guide:
- Drysdale, D. An Introduction to Fire Dynamics, 3rd Edition (John Wiley & Sons, 2011) — the textbook of the field.
- Hurley, M. J. et al., eds. SFPE Handbook of Fire Protection Engineering, 5th Edition (Springer, 2015).
- Chemistry LibreTexts — Combustion Reactions.
- Wikipedia — Combustion.
- Defenders of Wildlife — Fire-adapted species.
- ASU Ask A Biologist — Do animals survive forest fires?
- NASA — Combustion research in microgravity.
- True North Gear — What are fire whirls and fire tornadoes?
Cite this article
Dinh, D. C. (2026, April 14). What Is Fire? The Simple, Complete Guide Anyone Can Follow. PyroRisk. https://pyrorisk.net/blog/what-is-fire/
Recent posts
Featured posts
Categories
Comments
Related posts

The Science of Fire: What You Think You Know Is Wrong
Explore the science of fire — from radical chain reactions and flame anatomy to flashover physics and why modern homes burn 7x faster than 1950s ones.

The Science of Pyrolysis: How Heat Unmakes Everything
Pyrolysis — heat breaking molecular bonds without oxygen — drives wildfires, battery thermal runaway, biochar, plastic recycling, and even coffee roasting.
Cone Calorimeter 101: HRR, THR, and MARHE Explained
What the cone calorimeter measures — heat release rate, total heat release, and MARHE — and why each one matters for fire safety.